by Andrea Menarini
(click on photos to enlarge image)
SOLID SILVER OR SILVERPLATE?
Solid silver object usually bear hallmarks to testify the
silver fineness of the metal. In most cases, also silverplate
items, besides the manufacturer's trademark, present well known
abbreviations or names to distinguish the metal used in the
plating process (EPNS, EPBM, M�tal Blanc, Alpaka, etc.).
This is the norm, but what to do when we find a piece unmarked
or with a "fantasy" or unknown mark? How to distinguish solid
silver from silverplate?
Silverplate is the term commonly used to identify an object made
of base metal (white metal, bronze, copper, etc.) on which a
thin layer of silver was transferred by a process of electro
deposition. The silver coat is extremely thin and rarely exceeds
50 microns (5 hundredths of a millimetre). Often, the repeated
use and the cleaning remove the thin layer of silver bringing to
light the underlying metal (particularly in the protruding parts
of the piece). Obviously, in this case, to identify the metal as
silverplate is a simple task.
Otherwise, you can use one of the chemical reagents available on
the market to test the silver quality.
It consists of a strong acid (nitric or sulphuric acid) mixed in
appropriate proportions with a solution of potassium dichromate.
A drop of this yellow color reagent, placed on a silver surface,
causes a chemical reaction of bright red color (silver chromate).
This reaction happens only when the surface is solid silver. The
reagent can not detect the fineness; pure silver (999%) and its
alloys (925, 800) react at about the same way (only slight
differences in speed and intensity of the reaction will be
noticed by experienced eyes).
With this simple test you will be able recognize solid silver
from other metals as nickel silver, white metal and so on.
As mentioned above, however, the outer surface of a silverplate
object is silver in all its aspects, including the chemical
composition.
Therefore, depositing the liquid on the outer surface of an item,
the reaction will be anyway red, whether it is or is not solid
silver.
What to do to obtain a decisive result?
It is necessary to remove a thin layer of metal on a hidden part
of the object, as under the base or in an other area that does
not compromise the aesthetic of the piece (consider that silver
plate has a subtle layer of silver, thus the part to remove can
be minimal).
After this operation, and having thoroughly cleaned the
uncovered part, place a drop of reagent on the surface. The red
color will confirm that it is solid silver, no reaction or a
greenish color will reveal that it is silverplate.
In case of doubt, repeat the test after a thorough cleaning. The
procedure is slightly invasive (a small layer of metal will be
removed), but working in the right place the object will not
suffer perceptible damage. Moreover, on solid silver pieces, an
accurate sanding and polishing will remove any trace of the test
(but this is not possible in silverplate items).
For cutlery (or other pieces with no "hidden" parts) the test
with chemical reagent is not possible without damaging the
object.
In this case, you can make a proof with a "stone of comparison"
(can be purchased in the same stores that provide the reagent).
Two scrapes on the same surface of the metal will be necessary.
The first strip can be misleading, because it may contain parts
of the silvered surface; the second should contain only the
underlying metal.
Place a drop of reagent on the strip and, with the procedure
previously described, the presence or absence of the red
reaction will confirm the quality of the metal. This procedure
is apparently less invasive. Nevertheless, it leaves a little
smear on the metal surface.
Now, some useful suggestions for people interested to the matter.
Do not forget that the reagent is a chemical product, corrosive
and toxic. It is necessary to keep the product out of reach of
children, to handle it with care and to avoid skin contact and
ingestion.
To obtain reliable results on the chemical reaction, the test
requires an absolutely clean surface and the use of the reagent
"as sold", without any addition, dilution or contamination.

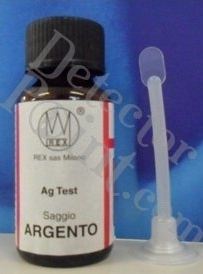
|
This product is readily available for a few
dollars in the stores dealing with materials for
gold and silvesmiths and on the Internet (search for
liquid silver test, testing silver, reactif argent,
silver testing solution, etc.).
A great variety of packages is available. Some
companies (in the USA) use a package similar to the
eye drops bottle (by far the most convenient,
allowing to pour the drop directly on the piece).
Other companies sell the reagent in a phial of thin
glass (once broken, is necessary to transfer the
liquid to a suitable container). Another option (found
in Italy) uses a bottle with a plastic spoon to pick
up the liquid.
It is absolutely necessary to pick up the liquid
with a tool made a material that does not react with
acids, such as glass or plastic, never metal.
It is also required a thorough cleaning of the spoon
after its use, to avoid the introduction of
contaminants into the liquid.
|
Following these suggestions you should be able to test your
objects and ascertain if they are or are not made of solid
silver.
The situation is different if the object is not yours, and you
wish to submit to test an object you intend to buy.
In my opinion, the test is not necessary if you are negotiating
with a professional dealer who issues written guarantee of the
quality of the metal.
More caution is needed if you are dealing with a non
professional seller on a flea market or a garage sale. In this
case, arranged the price and before paying it, you can request
to test the piece presented as solid silver.
In my experience, is rare that the seller objects to your
request if you declare that, in case of refusal, you give up the
purchase.
Great care is necessary in the transport of the reagent, as a
minimum leakage may cause personal injury or ruin your clothes.
Usually, I pour the product in an empty eye drops container,
thoroughly washed and dried.
To prevent drops oozing (due to temperature and / or pressure
variation) is necessary to seal the bottle in another envelope
such as a plastic bag.
NOTE: In this writing I describe the use of the potassium
dichromate, which is probably the most used reagent in Europe.
Elsewhere, for example in the East and in the United States, a
common alternative is pure nitric acid, of which I have no
direct experience. This acid gives reactions of different colors:
a creamy color for sterling silver or other higher fineness,
grey for 800 silver and light green in various shades as the
title goes down. The advantage of this system is the approximate
indication of the alloy title.
It would be interesting to obtain information about this test by
someone well acquainted with the matter.
|
|

